|
2/12/2024 0 Comments Atomic structure worksheet answer
The lightest element is Hydrogen its atomic weight is 1 and that is why it’s found in the prime left nook of the periodic table.One was john dalton also bring you the idea and lewis dot is the association of empty atoms of matter could be closed if they’re accountable for?.Historically, however, relative atomic masses have been used by scientists making an attempt to organise the … This Atomic Structure Worksheet with reply key PDF offers your college students an excellent training in how atoms work. The fashionable periodic table lists the weather in order of increasing atomic quantity. Atoms of the identical component that have totally different numbers of neutrons are … Chemists have all the time looked for methods of arranging the elements to replicate the similarities between their properties. The unit of measurement used for atomic particles is the. Identify the component that corresponds to every of the next electron configurations. You will check your data once again utilizing all that you have realized. When atoms mix, they create individual compounds which may be a half of the universe. They are the constructing blocks of all chemical structures. The significance of atoms is that without them, nothing may exist. Hydrogen is in water and comprises sixty-one p.c of the human body’s atoms. What contribution did these scientists make to atomic models of the atom? LEWIS STRUCTURES PRACTICE WORKSHEET Draw the Lewis Structures for every of the following molecules. What is the atomic massmass variety of the atom within the diagram above. All the mass of an atom is made up with these particles. And finally, the charge of an atom is determined by the number of protons and electrons in the atom.Atomic Structure Worksheet Answer Key. The mass number of an atom is found by adding together the number of protons and neutrons. The number of electrons will determine how atoms interact with one another and decide if the atom as a whole is positive, negative, or neutral. 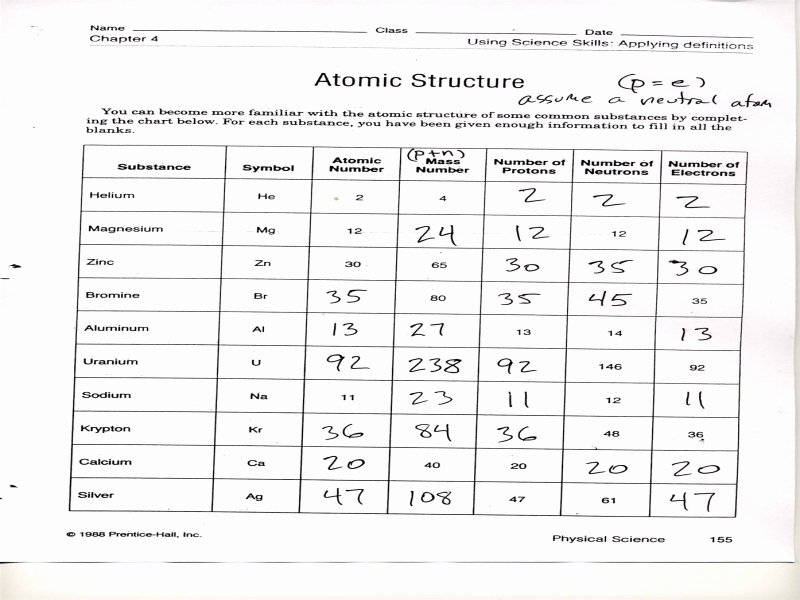
The charge of an atom is calculated based on the difference between the number of protons in the nucleus and the number of electrons orbiting the nucleus.Īn atom is made up of protons, electrons, and neutrons. The periodic table arranges atoms in increasing atomic number. Atoms have an atomic number, which is the number of protons in the nucleus. Now that we have talked about the different parts of the atom, let’s summarize a few properties of atoms. Water is made up of two hydrogen atoms and one oxygen atom. Atoms can share electrons to form molecules, which are particles made up of many atoms. The electrons determine how atoms interact with each other. With fewer electrons than protons, the atom will have a positive charge. If the atom has more electrons than protons, its charge will be negative. If the number of electrons is equal to the number of protons, the overall electric charge of the atom is neutral. The electrons are attracted to the positive nucleus, but they can escape their orbit by an outside force.Ītoms have a certain number of electrons orbiting the nucleus. Orbiting the atom’s positively charged center are particles with a negative charge called electrons. This gives us names like Carbon-12 or Carbon-14, which are types of carbon atoms used in carbon dating! To find the mass number of an element you add the number of protons and neutrons together, so protons + neutrons = mass number. Usually, a stable atom has an equal number of neutrons and protons, but there are exceptions. In general, an atom will have a specific number of neutrons in the nucleus, meaning the atom won’t lose or gain any neutrons for a very long time. For example, hydrogen is an element with one proton in the nucleus and carbon is an element with 6 protons. A group of atoms that all have the exact same number of protons is called an element. Protons are very small, positively-charged particles, and neutrons are neutral particles that have no charge.Ītoms can have just one proton or they can have multiple. 
What is an atom?Īt the very center of an atom is the nucleus, which is made up of small particles called protons and neutrons. Atoms are the building blocks of the universe they make up everything you see around you.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
